What Is Saponification?
Saponification is a process that converts fats, oils, or lipids (the acid) into soap by combining them with Sodium Hydroxide (the base). The chemical reaction relies on friction and self-generated heat. Through saponification, the acid and the base are neutralized. One of our favorite soap formulas is as follows: Combine organic olive oil, organic coconut oil, organic RSPO palm oil, and organic shea butter with sodium hydroxide. Knowing the proportions of acid to base needed for saponification is the key. We determine this by knowing the SAP value of each oil (acid) in the formula (there are charts for this). The SAP value is the amount of sodium hydroxide needed to saponify a particular oil completely. Each type of oil, whether vegetable or animal, has a different SAP value based on its molecular structure.
How Saponification Works
Once the proper amount of acid and base are combined, the chemical reaction begins, but this is not where our soapmaking ends. As we stir, the oils go from translucent to opaque. Then, we look for a trace. A trace is a telltale sign that our cold-process soap has reached a point in saponification where we can start to incorporate additives. We know our soap has traced when the spatula leaves a noticeable trail when drizzled.
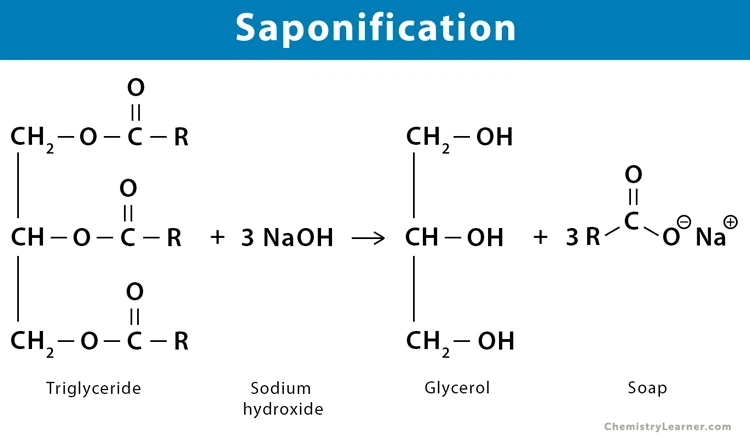
Global Reaction (One‑Liner)
Fat/Oil (Triglyceride) + NaOH or KOH → Soap (Sodium or Potassium Salt of Fatty Acid) + Glycerin + Heat
Industrial vs. Artisan Approaches
One‑step industrial batch: fats + lye under steam, then salt‑out.
Two‑step continuous process: fat splitting → free fatty acids → neutralization.
Artisan cold/hot process: completed in‑mold or in‑pot, retaining glycerin for skin feel.
Exothermic Curve
Even "cold‑process" soap self‑heats to 90 – 120 °F (32 – 49 °C); managing that mini‑thermal spike keeps colors and fragrances true.
The Key Reactants
| Component | Why it Matters | Pro Tips |
|---|---|---|
| Oils & Butters | Fatty‑acid profile governs hardness, lather, conditioning. | Blend saturated (coconut, palm) with unsaturated (olive, sweet almond) for balanced bars. |
| Alkali (NaOH vs KOH) | Sodium yields hard bars; potassium produces soft/liquid soap. | Use 99 % pure pearls or flakes; weigh to 0.1 g accuracy. |
| Water Phase | Dissolves lye, moderates heat. | "Water discount" (5 – 15 %) accelerates cure and hard‑ness. |
| Additives | Salt for hardness, sugar for bubbles, sodium lactate for unmolding. | Add after light trace to avoid premature thickening. |
What happens during saponification?
During saponification, the fats/oils release their glycerol, which becomes glycerin. The hydroxide ion (OH) from the lye combines with the fatty acids from the fats/oils and becomes soap. The end result of good handmade soap is a balanced bar that contains both soap and glycerin. When soap is handmade through the cold process method, the glycerin is in balance in the soap. In commercial soaps, the glycerin is often removed because it can be sold for a higher profit than with it left in the soap.
It's important to note that saponification is an exothermic reaction which means it gives off heat.
How Different Soap‑Making Methods Affect Saponification
| Method | When Saponification Completes | Pros | Typical Cure Time |
|---|---|---|---|
| Cold Process | In‑mold over 24 h | Silky texture, creative pours | 4 – 6 weeks |
| Hot Process | In‑pot within 1–2 h | Ready to use faster | 1 – 3 days |
| Liquid Soap (KOH) | Multi‑stage paste dilution | Crystal‑clear gels | No cure |
| Melt & Pour | Pre‑saponified base | Zero lye handling | Immediate |
Variables That Shape Soap Quality
Temperature Window: 85 – 115 °F (29 – 46 °C) is the sweet spot for most designs.
Trace Consistency: From light pudding to thick custard; determines swirl success.
Water Discount: Less water = faster hardening but shorter swirl time.
Equipment: Stainless or HDPE only; aluminum reacts with lye.
Additives: Clays thicken quickly; certain fragrance oils accelerate (a.k.a. "seize").
Troubleshooting Common Saponification Issues
| Symptom | Likely Cause | Fix |
|---|---|---|
| Lye Pockets / Zaps Tongue | Incomplete mixing, false trace | Re‑batch with 1–2 % water, heat gently. |
| False vs. True Trace | Pure hard oils + low temp mimic thickness | Stick‑blend a few more seconds and check fluidity. |
| Seizing / Soap on a Stick | Fragrance acceleration, high temps | Work cooler; pre‑blend fragrance in oils. |
| Overheating Cracks | Thick mold insulation | Vent the top, spritz with isopropyl alcohol. |
Frequently Asked Questions
1. Is saponification necessary to make real soap?
Yes. Saponification is the chemical process that defines true soap-it transforms fats or oils and an alkali (like lye) into soap and glycerin. Without this reaction, you're not making real soap but simply mixing surfactants or detergents.
2. What does "trace" mean in saponification?
Trace is the point in soap making when the oil and lye solution has emulsified enough to leave visible trails on the surface when stirred. It's the sign that saponification has begun and you can safely pour the mixture into molds.
3. Does soap continue to saponify during curing?
Yes, though most of the reaction finishes in 24–48 hours, subtle changes continue during the 4–6 week curing phase. This allows excess water to evaporate, improves lather, and completes any lingering saponification.
4. Can I make soap without lye?
Not really. While you can use a pre-made "melt and pour" soap base (where lye has already been used), all true soap is made with lye at some stage. There's no soap without saponification, and there's no saponification without lye.
5. Why does saponification produce heat?
Saponification is an exothermic reaction, meaning it releases heat as bonds break and form between fatty acids and lye. This natural heat is especially noticeable in cold process soap and helps drive the reaction to completion.