What is the surface tension of water?
Surface tension is the tendency of liquid resting surfaces to shrink into the minimum surface area possible. Surface tension allows objects with a higher density than water such as razor blades and insects (e.g. water striders) to float on a water surface without becoming even partly submerged.
At liquid-air interfaces, surface tension results from the greater attraction of liquid molecules to each other (due to cohesion) than to the molecules in the air (due to adhesion).
There are two primary mechanisms in play. One is an inward force on the surface molecules causing the liquid to contract. Second is a tangential force parallel to the surface of the liquid. This tangential force is generally referred to as the surface tension. The net effect is the liquid behaves as if its surface were covered with a stretched elastic membrane. But this analogy must not be taken too far as the tension in an elastic membrane depends on the amount of deformation of the membrane while surface tension is an inherent property of the liquid–air or liquid-vapor interface.
Because of the relatively high attraction of water molecules to each other through a web of hydrogen bonds, water has a higher surface tension (72.8 millinewtons (mN) per meter at 20 °C) than most other liquids. Surface tension is an important factor in the phenomenon of capillarity.
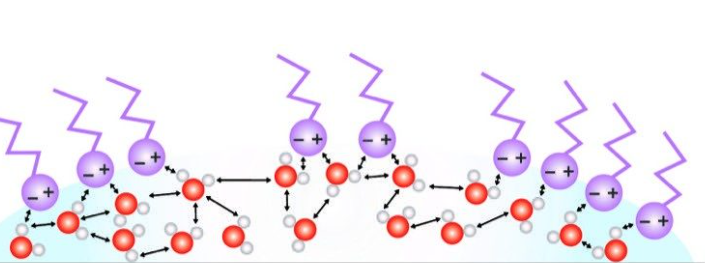
Why does soap break water surface tension?
Soap breaks the surface tension of water due to its molecular structure, which contains both hydrophobic (water-repelling) and hydrophilic (water-attracting) ends. Water molecules at the surface create a "skin" due to cohesive forces between them, leading to high surface tension.
When soap is added to water, the hydrophobic ends of the soap molecules are repelled by water and push their way to the surface, while the hydrophilic ends remain in the water. This disrupts the cohesive forces between water molecules at the surface, reducing the surface tension. As a result, the water becomes less cohesive and spreads out more easily, allowing it to wet surfaces better.





